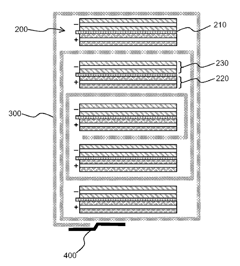
Ceramic-filled Separators from Porous Power

Unlike dense ceramic coatings applied as a separate layer on competing separators, PPT’s products contain ceramic particles that are thoroughly distributed throughout their chemically inert PVDF polymer. The particles reside primarily in the pore walls, leaving a large and uniform open pore structure that enables ions to pass through with less resistance and less heat generation. * More power in high-current applications * Reduced separator shrinkage * Increased thermal stability with less cell degradation * Extended cycle life * Higher bake-out and operating temperatures * Improved puncture and abuse resistance Although, the application of such separator may improve safety, however according to presented test data it will have minimal effect, or not significant. Depends on battery package, it may have serious explosion (due to electrolyte flammability) at much lower temperature.